What purpose do organoids serve?
© Saba Rezakhani, Lütolf lab /EPFL
ORGANOIDS FOLDER 2/3 – Three researchers tell us about their projects. Their research topics are totally different but all involve organoids.
Modeling an intestine
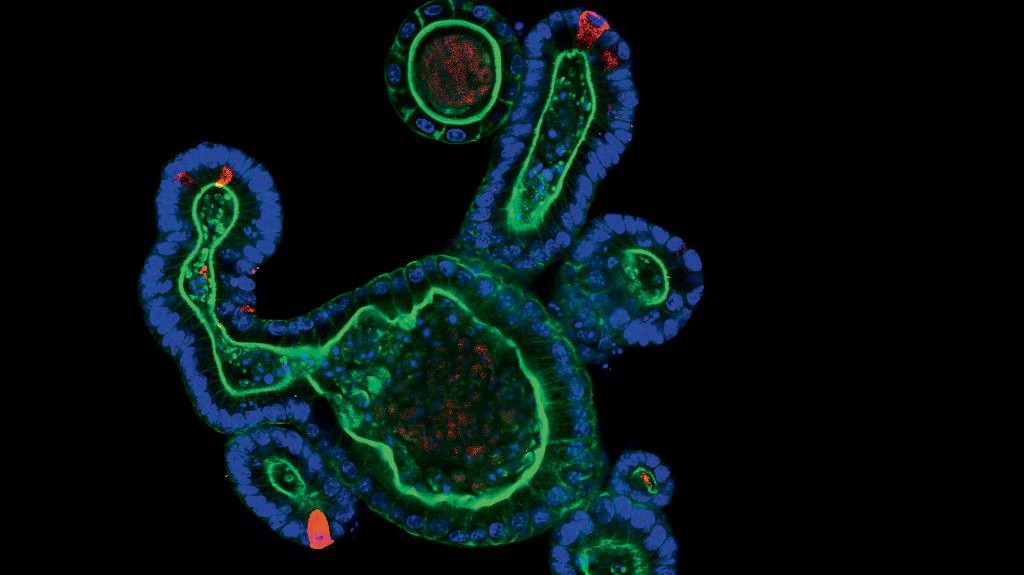
 Professor Matthias Lütolf from EPFL’s Interfaculty Institute of Bioengineering (IBI) is working on modeling micro-intestines. His laboratory is looking to better grasp how stem cells decide which tissue to form and how that process is influenced by the signals from their environment.
Professor Matthias Lütolf from EPFL’s Interfaculty Institute of Bioengineering (IBI) is working on modeling micro-intestines. His laboratory is looking to better grasp how stem cells decide which tissue to form and how that process is influenced by the signals from their environment.
Using epithelial stem cells from the intestine, he has created a small structure that mimics the surface of the intestine. “We can see primitive villi containing differentiated cells that absorb nutrients just like a real intestine, as well as crypts containing stem cells. It’s amazing!” says Lütolf. “But these organoids of intestines are shaped like little balls. So in the lab, we’re working on making them more realistic by controlling the environment outside the cells and integrating additional cells that are important for the organ, such as those from our immune system.” This work, which requires in-depth know-how of microengineering and of how to produce sophisticated biomaterials, is at the crossroads between biology and engineering.
“We’ve developed gels that guide cells so that they grow into certain shapes by creating cavities that restrict the space available. We can also adjust the physical and biochemical properties of the gel to manipulate the cell’s shape,” he explains. “The aim is to create a larger tube-shaped organoid that more closely resembles an intestine. We’ll then be able to get inside to insert, for example, intestinal bacteria and study many more physiological phenomena.”
The researchers have just teamed up with Professor George Coukos, head of CHUV’s oncology department. Using biopsies from cancer patients at CHUV, they’ve created tumor organoids that they are trying to make more realistic by reproducing their microenvironment – the normal cells surrounding the tumor. These complex organoids can then be compared with the tumors still present in the patient. The aim is to analyze whether the effect of an anti-cancer drug is similar in both cases.
Professor Lütolf’s work has won recognition from Europe’s top life scientists, and he became a member of the European Molecular Biology Organization (EMBO) in 2018.
Finding the right drug
 Professor Mark Rubin from the University of Bern and Inselspital, Bern’s university hospital, has been using tumor organoids in a clinical setting in order to model cancer treatments. He is one of Switzerland’s most experienced and well-known experts in this field.
Professor Mark Rubin from the University of Bern and Inselspital, Bern’s university hospital, has been using tumor organoids in a clinical setting in order to model cancer treatments. He is one of Switzerland’s most experienced and well-known experts in this field.
Professor Rubin is working to develop precise cancer treatments. The goal is to identify effective therapies for each individual patient, since not all tumors react in the same way to the same drugs, and treatment has to be adapted to how the tumor evolves over time.
“Using the cells taken from a patient’s tumor, it is possible to grow a whole collection of spherical organoids,” explains Rubin. “We can then test various drugs on each of the micro-tumors. We’ve demonstrated that this technique is reliable enough to predict how a patient will react to a given drug and to determine which will be the best option for the next treatment phase. This is an important step because it was previously thought that this had to be verified in vivo by grafting the tumor onto an animal, a process known as a xenograft.”
Studying embryo development
 Professor Denis Duboule from EPFL’s Swiss Institute for Experimental Cancer Research (ISREC-SV) and the University of Geneva, is interested in the development of vertebrates. He began his research in this area more than 30 years ago and started using organoids of early mouse embryos in recent years.
Professor Denis Duboule from EPFL’s Swiss Institute for Experimental Cancer Research (ISREC-SV) and the University of Geneva, is interested in the development of vertebrates. He began his research in this area more than 30 years ago and started using organoids of early mouse embryos in recent years.
Why do humans have 12 thoracic vertebrates with ribs and not 11 or 13? What determines where our organs are located in our thorax? These are just some of the questions that embryology is trying to answer. “To study the genes that control this structuring process, we’re using organoids of mouse embryonic stem cells,” says Duboule. “They’re known as gastruloids, which are present in a phase early in embryonic development, before a major reorganization of the cells known as gastrulation occurs.” These gastruloids are not embryos, but rather structures that are organized in a similar way to embryos under certain culture conditions. The cells grow, but not in the right direction, which means that the subsequent development phases – such as the formation of organs like the liver or kidneys – will never be triggered.
Gastruloids alone are not enough to study embryo development: “It’s just one technique that can help us answer certain very specific questions,” says Duboule. “It won’t replace the study of real embryos. And the results obtained have to be verified on actual embryos anyway.” But gastruloids nevertheless offer some advantages: “They enable us to see the genetic activity with our own eyes in real time! We just have to insert a fluorescent protein into the gene we want to observe. This saves us considerable time compared with our work on mice. What used to take a year now takes just two months. And it’s reduced the need for animal testing as well.”
Photos credits: © Alban Kakulya, John Abbott and Alain Herzog.
Find all the articles of the folder dedicated to organoids:
1 - Organoids – mimicking our organs in the lab
2 - What purpose do organoids serve?