Uncovering the biology of a painful and disfiguring pediatric disease
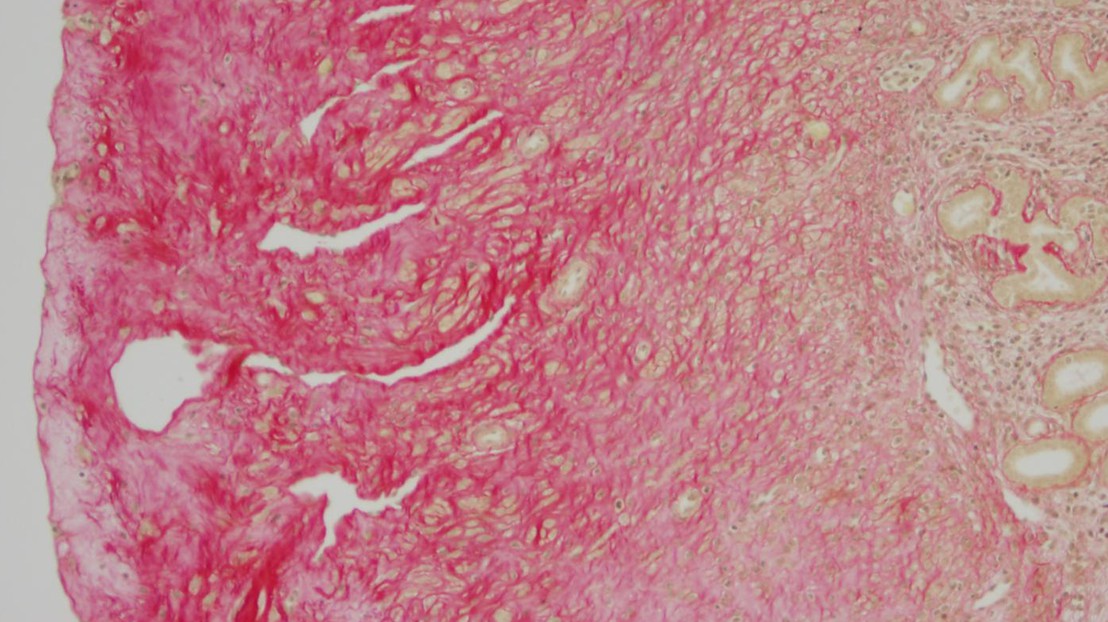
Accumulation of extracellular material in mice uterus. © EPFL
EPFL scientists have identified the biological mechanism behind the painful, potentially lethal, disfiguring Hyaline Fibromatosis Syndrome.
Hyaline Fibromatosis Syndrome (HFS) is a rare but severe genetic disease that affects babies, children, and adults. A glassy substance called hyaline accumulates in the skin and various organs of patients, causing painful deformities and may lead to early death. The disease is caused by gene mutations that destroy the function of a protein whose physiological function is little understood. EPFL scientists have now studied and characterized its function for the first time, showing that it binds and controls the body levels of an important type of collagen. The work is published in Nature Communications.
HFS is caused by a mutation of a gene called CMG2, also abbreviated as ANTXR2 because it renders cells sensitive to anthrax infections. The gene produces a protein that is embedded in the membrane of cells, and is known to bind anthrax toxins internalizing them into the cell where they exert their action. This is however not the physiological function of this protein.
The HFS mutation of the gene causes the protein to lose its function. A hallmark of the disease is the accumulation of collagen in large nodules under the skin. Collagen is the most abundant protein in mammals, found in skin, cartilage, blood vessels, and even teeth. There are many different types of collagen, which surround cells and support the body’s various tissues and organs by forming elastic or rigid structural networks.
The lab of Gisou Van der Goot at EPFL has now shown for the first time that the CMG2 protein interacts with a type of collagen called collagen VI. Normally, CMG2 binds collagen VI and, when there is too much, internalizes it into the cell’s lysosomes for degradation. The study suggests that, in HFS, the mutation of the CMG2 gene disrupts the function of the protein, leading to accumulation of collagen VI in various parts of the body.
Collagen VI is a type of collagen that recent studies have found in multiple sites in the body, such as the heart, neurons, immune cells, tendons, lungs, and the skin. Depending on the type of tissue, it plays different protective and mechanical roles, but it has also been shown to be involved in a number of diseases, including the growth of cancer tumors.
The EPFL researchers studied HFS nodules in mouse models as well as a human patient. The female HFS mice characteristically suffer from accumulation of collagen in the uterus, which makes them sterile. The human patient had nodules at various body sites including the scalp and behind the ear. Studying tissue samples, the researchers found that the nodules in both mice and human were rich in collagen VI.
Wanting to understand the relationship between CMG2 and collagen VI, the team developed double-knockout mice in which the genes for both proteins — CMG2 and collagen VI — had been deleted. Previous studies have shown that CMG2-knockout mice are infertile due to uterine nodules that render them infertile. But remarkably, the scientists found that when the gene for collagen VI was deleted too, the mice regained their fertility, confirming that CMG2 and collagen VI are somehow connected in HFS.
Further studies showed that CMG2 physically binds collagen VI through a specialized domain. The CMG2 protein then acts as a receptor, sending a signal into the cell, causing collagen VI to be internalized and degraded inside the cell’s lysosomes.
The study reveals a major physiological function for CMG2 and demonstrates its interaction with collagen VI. This interaction explains how major HSF symptoms arise when mutation of the CMG2 gene disrupts the ability of the CMG2 protein to control the levels of collagen VI, which then over-accumulates and produces the painful and disfiguring symptoms of the disease.
This study was led by EPFL’s Global Health Institute in collaboration with the University of Bern, the Medical University of Innsbruck, the University of Freiburg, the Centre hospitalier universitaire vaudois (CHUV), and the University of Padova. It was funded by the Swiss National Science Foundation (SNSF), the Gelu Foundation, theFrancis and Marie-France Minkoff Foundation, the Solis Foundation, and the Associazione ISI.
Reference
Jérôme Bürgi, Béatrice Kunz, Laurence Abrami, Julie Deuquet, Alessandra Piersigilli, Sabine Scholl-Bürgi, Ekkehart Lausch, Sheila Unger, Andrea Superti-Furga, Paolo Bonaldo, F. Gisou van der Goot. CMG2/ANTXR2 regulates extracellular for collagen VI which accumulates in Hyaline Fibromatosis Syndrome.Nature Communications