The “switch” that keeps the immune system from attacking the body
A dendritic cell, one of the cells of the innate immune system where cGAS plays a major role ©EPFL/iStock photos (Design Cells)
Scientists at EPFL uncover the mechanism by which cells mark the protein cGAS for degradation, which is critical in preventing the immune system from mistakenly attacking the body's own tissues.
A microscopic battle rages in our bodies, as our cells constantly fend off invaders through our immune system, a complex system of cells and proteins designed to protect us from harmful pathogens. One of its central components is the enzyme cyclic GMP-AMP synthase (cGAS), which acts as a sentinel, detecting foreign DNA and initiating an immune response.
However, the immune system requires precise regulation to prevent cGAS from mistakenly attacking the body's own tissues, leading to autoimmune disorders, which now affect about 10% of the global population.
Previous studies have revealed a little of how this happens. During cell division – mitosis – the membrane that protects the cell’s nucleus, the nuclear envelope, breaks down and cGAS quickly relocates into the nucleus. There, it attaches itself to nucleosomes – the basic structural unit of DNA packaging in the cell – and becomes covered by another protein called BAF.
All this ensures that cGAS stays inactive and fixed in place, and does not mistakenly interact with the cell's own DNA. It represents a sophisticated balance between immune readiness and protecting the integrity of the cell’s genome. The question is, how does the cell coordinate this with its other everyday functions?
A new study from the group of Andrea Ablasser at EPFL sheds light on how cGAS is regulated, especially during the critical phase of cell division known as mitosis. The study is published in Nature.
The team used advanced imaging and molecular techniques to observe how cGAS is selectively broken down in the nucleus, preventing it from mistakenly responding to the cell's own DNA. They found that the process is mediated by a protein complex known as CRL5–SPSB3, which recognizes a specific motif in cGAS and tags it in the nucleus for destruction. Using structural biology, biochemistry, and cell biology, the researchers visualized the interactions between cGAS and the protein complex at the atomic level.
Specifically, CRL5–SPSB3 adds a protein called ubiquitin to cGAS. Ubiquitin – as the name suggests – is found ubiquitously across eukaryotic cells and one of its functions is to mark other proteins for death. The ubiquitination of cGAS also marks it for destruction, effectively inactivating the sentinel once the threat of an invader has been neutralized.
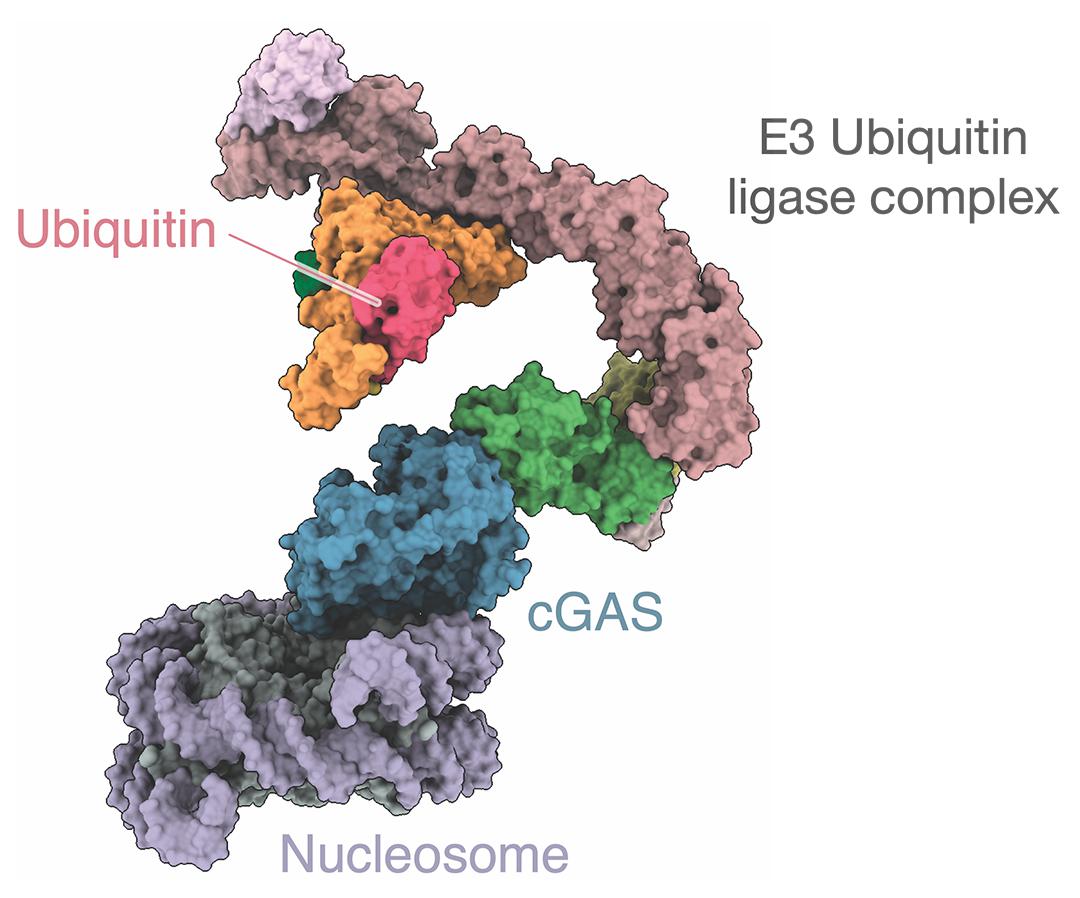
By elucidating the structure of the cGAS-SPSB3 complex, the study maps out how cGAS is regulated within the nucleus of cells, highlighting the sophistication of the immune system`s regulatory networks.
The implications also extend beyond basic science, allowing scientists to explore new strategies for treating diseases where the immune system is either overactive, such as in autoimmune diseases, or underactive, as in cases of chronic infections or cancer. For example, modulating cGAS activity could potentially enhance the effectiveness of cancer immunotherapies or provide new approaches to preventing autoimmune conditions.
Other contributors
EPFL Institute for Cancer Research (ISREC)
Swiss National Science Foundation
Swiss Cancer League
Dr. Josef Steiner Cancer Foundation
European Molecular Biology Organization (EMBO)
Pengbiao Xu, Ying Liu, Chong Liu, Baptiste Guey, Lingyun Li, Pauline Melenec, Jonathan Ricci, Andrea Ablasser. The CRL5–SPSB3 ubiquitin ligase targets nuclear cGAS for degradation. Nature 28 February 2024. DOI: 10.1038/s41586-024-07112-w