Solving a Parkinson's disease puzzle through protein design
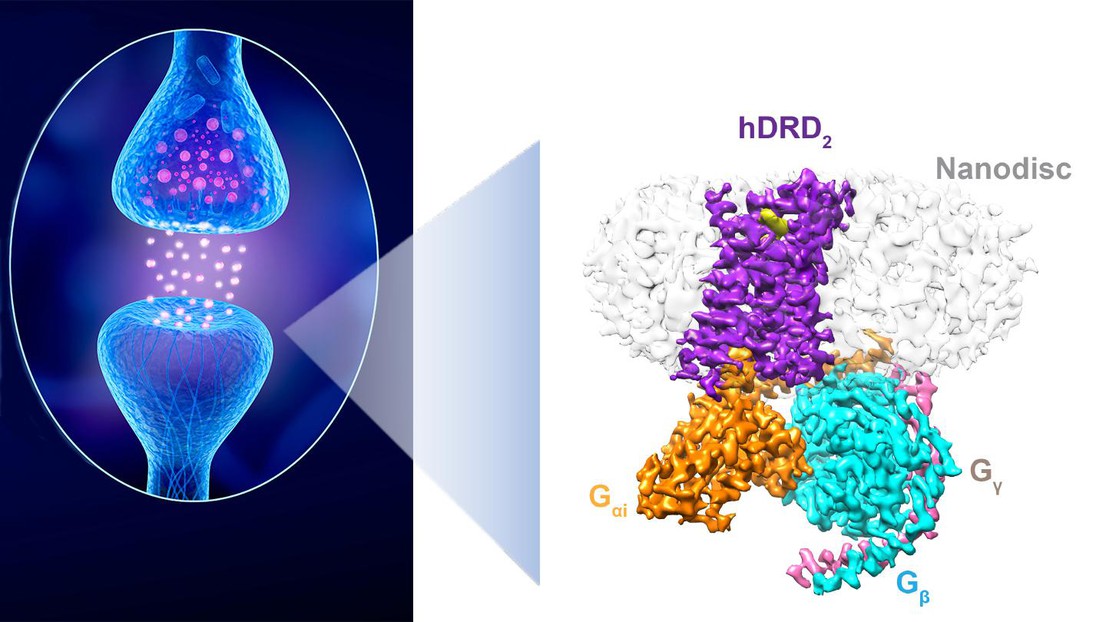
A dopaminergic synapse between a neuron releasing dopamine and a neuron sensing the neurotransmitter. Right: The structure of a D2 receptor in “action”. Credit: iStock and P. Barth (EPFL)
EPFL researchers, in collaboration with UTSW and UCSD scientists, have developed a computational protein design approach, and used it to obtain the first ever high-resolution structure of an activated dopamine receptor in its natural cell membrane environment. The breakthrough will open up a new dimension in drug discovery for Parkinson’s disease and perhaps other disorders.
Dopamine is a neurotransmitter involved in everything from higher cognitive functions to motor control, motivation, arousal, reinforcement, and sexual gratification, the receptors it acts on have been a longstanding target for treating disorders like Parkinson’s disease, which is caused by the degeneration of dopamine-using neurons that control movement.
The problem is that for at least two decades, no-one has been able to “see” what a dopamine receptor looks like when it is activated by dopamine – at least not in high enough resolution to offer avenues for designing drugs that can target the receptors effectively.
In a major collaborative study published in Nature, scientists from the lab of Patrick Barth at EPFL, with colleagues at UTSW and UCSD have now worked out the high-resolution structure of an activated form of a dopamine receptor in a native lipid membrane environment. “The native receptor is so misbehaved and its active form so transient that attempts at observing the receptor structure ‘in action’ have failed so far,” says Barth.
The way the scientists solved the problem was by combining state-of-the art computational allosteric and de novo protein design approaches developed by Barth’s group allowing the researchers to engineer a highly stable but activated dopamine receptor whose structure they could then study and solve.
The EPFL team created a receptor with artificial building blocks such as activating switches and de novo binding sites, which replaced unstable, structurally disordered, and inactivating regions of the native receptor.
“This hybrid functional/de novo computational protein design approach is powerful, as it enabled us to create a receptor with considerably enhanced activity and stability while recapitulating key native functionalities such as dopamine-mediated intracellular signaling and binding,” says Barth.
The success was also made possible by using high-end lipid reconstitution techniques and cryo-electron microscopy, overcoming obstacles in previous studies that attempted to determine the receptor’s structure using X-ray crystallography and by keeping the receptor inside detergents.
The problem is that detergents are very poor mimics of the cell’s lipid membranes where receptors like the dopamine one are naturally located. Also, detergents have a reputation of distorting and even inactivating receptors, which doesn’t help when attempting to see what they look like in action. “This represents the first atomic-level membrane-receptor structure determined in a native lipid bilayer,” says Barth.
“The breakthrough will enable improved drug discovery efforts against, for example, Parkinson disease,” he adds. “But it also sets the stage for broadly applying functional and de novo protein design approaches to accelerate the structure determination of challenging protein targets and create proteins with novel functions for a broad range of therapeutic and biotechnological applications.”
Edward Mallinckrodt, Jr. Foundation
Welch Foundation
EPFL
Ludwig Institute for Cancer research
UTSW
CPRIT
National Institutes of Health
Jie Yin, Kuang-Yui M. Chen, Mary J. Clark, Mahdi Hijazi, Punita Kumari, Xiao-chen Bai, Roger K. Sunahara, Patrick Barth, Daniel M. Rosenbaum. Structure of a D2 dopamine receptor-G protein complex in a lipid membrane. Nature 11 June 2020. DOI: 10.1038/s41586-020-2379-5