Shedding light on electron trapping
Trapped Electrons ©Angewandte Chemie
Using a pioneering technique, EPFL scientists have shown how electrons move and get trapped in titanium dioxide, which is used in Grätzel cells, self-cleaning windows and water-purifying technologies.
Titanium dioxide (TiO2) is the material most used in photovoltaic dye-sensitized solar cells, as well as in photocatalytic applications, such self-cleaning glass. In both technologies, the underlying principle is the creation of electrical charges, electrons (negative) and holes (positive). However, the fate of these charges after their creation remains a fundamental question. Using a unique method, EPFL researchers have for the first time determined the nature of the electron traps in TiO2, which can significantly optimize its use in light-based technologies. Their work is published in Angewandte Chemie.
Converting light into electrical or chemical energy is one of the major goals of our time, with enormous implications on the future of mankind. Applications include photovoltaic devices like dye-sensitized solar cells (DSSC; Grätzel cells) and photocatalytic devices like self-cleaning windows and water-purification systems. Most devices are made of a crystalline form of TiO2 called anatase, which is often sensitized with a light-absorbing dye molecule that injects electrons into the TiO2 substrate.
In photovoltaic devices, the electrons are the main actors, while for photocatalysis, they reduce impurities that attach to a surface or the electron holes, and oxidize them by, respectively, adding or removing electrons from these impurities. Thus, the efficiency of photovoltaic and photocatalytic devices depends not only on the transport of charges in the TiO2 lattice, but also on the fact that they are trapped in the right place. In the case of electrons, the nature (location, geometry, etc.) of the traps and their lifetime, particularly at room temperature, has remained a mystery.
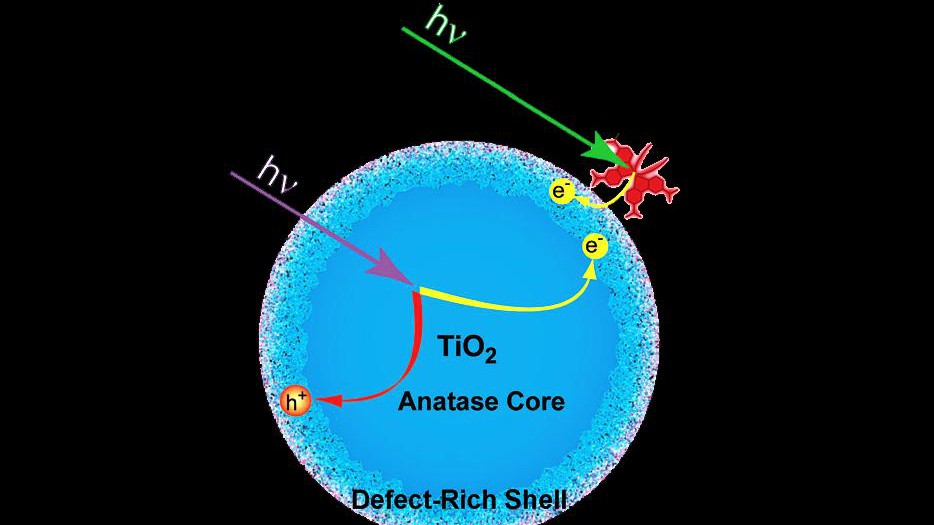
The group of Majed Chergui at EPFL has used time-resolved X-ray absorption spectroscopy (XAS) to study how electrons are trapped in the anatase (ordered) and amorphous (disordered) forms of TiO2 at room temperature. XAS is a powerful, element-specific technique for determining the electronic structure of atoms and the geometric structure around them. In this novel study, the researchers examined photo-excited bare and dye-sensitized TiO2 nanoparticles in colloidal solutions. Time-resolved XAS, which was pioneered in Chergui’s lab, allowed the researchers to follow electron movements on a 100 picosecond scale (10-10 sec) and to visualize the formation of Ti3+-type electron traps (a Ti atom of the lattice, with an additional electron on its outer shell).
The results show that in both bare and dye-sensitized anatase, electrons get trapped in the defect-rich surface shell of the TiO2 substrate; deep inside the shell in the case of bare anatase and on the outer surface in the case of dye-sensitized nanoparticles. For bare anatase nanoparticles, the electrons can travel through the regular lattice towards the surface, where they become trapped as soon as they encounter defects, which is the case deep inside the surface shell. The travel time of the electron is well below the time resolution of the experiment (100 ps). For dye-sensitized nanoparticles, the electrons are immediately trapped at the surface, because defects are dominant there.
The researchers found that in bare amorphous TiO2, the electrons become trapped in the disordered, defect-rich inner bulk of the substrate, while when a sensitizing dye is added on the TiO2, the electrons get trapped on its surface, as in the case of the anatase form. The researchers have determined the lifetime of the different electron traps in bare and dye sensitized TiO2, which span from nanoseconds to tens of nanoseconds.
This fundamental discovery demonstrates the power of time-resolved XAS to study a previously inaccessible phenomenon. In addition, clarifying the TiO2 electron-trapping dynamics has implications on the manufacturing of DSSCs and photocatalytic devices, as using the considerably cheaper amorphous (non-crystalline) TiO2 can achieve similar efficiencies in trapping electrons at the surface of the substrate.
“In the sense that you need long-lived charges at the surface and cheap materials for photocatalysis, use dye-sensitized amorphous TiO2”, says Chergui. “If you want to do charge-transport, you do need traps – you have to be careful, because mesoporous anatase contains a lot of defects at the surface, and that is where most electrons are going to get trapped.”
Reference
Rittman-Frank MH, Milne CJ, Rittmann J, Reinhard M, Penfold TJ, and Chergui M. Mapping of the photoinduced electron traps in TiO2 by picosecond X-ray absorption spectroscopy. Angewandte Chemie Intern. Ed. DOI:10.1002/anie.201310522