Self in spatial navigation and human grid cell activity
© 2022 EPFL
Hyuk-June Moon and colleagues report in a new paper, just published in Communications Biology, that the sense of self impacts spatial navigation and hexadirectional coding in human entorhinal cortex.
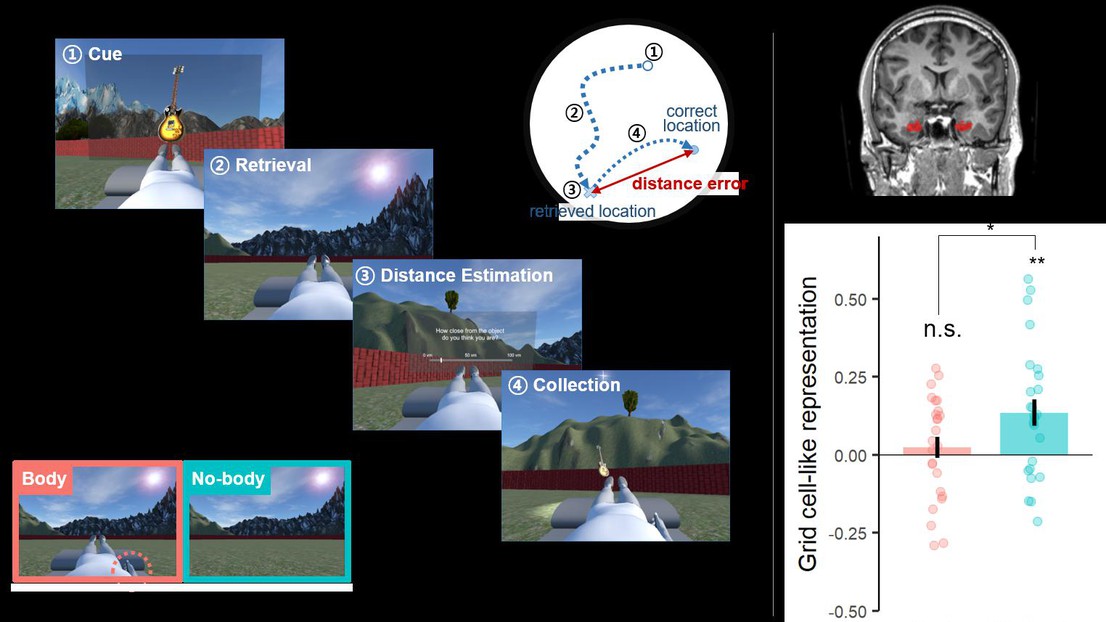
Grid cells in entorhinal cortex (EC) encode an individual’s location in space and rely on environmental cues and self-motion cues derived from the individual’s body. Body-derived signals are also primary signals for the sense of self or self-consciousness and are based on integrated sensorimotorsignals (proprioceptive, tactile, visual, motor) that have been shown to enhance self-centered processing. However, it is currently unknown whether such sensorimotor signals that modulate self-centered processing impact grid cells and spatial navigation.
In the recent paper, Moon et al. (2022) have developed a new immersive virtual reality (VR) technique that allows to integrate online manipulations of bodily signals to modulate the sense of self (with respect to an avatar) while subjects performed a spatial navigation task. Importantly, they also acquired fMRI data and detected grid cell-like activity in human entorhinal cortex during this task. Moon et al. report improved performance in spatial navigation and decreased GCLR in EC when participants navigated in VR when self-identifying with the avatar in VR. This decrease in entorhinal GCLR was associated with an increase in retrosplenial cortex activity, which was correlated with participants’ navigation performance. These are the first data that link self-centered processes to spatial navigation and to entorhinal as well as retrosplenial activity, highlighting the role of the sense of self for spatial navigation.
Supported by the Korean Government Scholarship Program for studyoverseas, the Korea Institute of Science and Technology (KIST) Institutional Program (2E31642), and the Bertarelli Foundation to H.-J.M. O.B. is supported by the Swiss National Science Foundation (No. 320030_188798) and by the Bertarelli Foundation. N.F. has received funding from the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation program (Grant 803122). Additional support was provided by the Fondation Campus Biotech Geneva (FCBG) - a foundation of the Swiss Federal Institute of Technology Lausanne (EPFL), the University of Geneva (UniGe), and the Hôpitaux Universitaires de Genève (HUG), the Institute of Translational Molecular Imaging (ITMI).