Congratulations to Petru for his JAC paper!
© 2026 EPFL
We cannot address what we don't understand, so do want to learn more about what causes copper instability during CO2 electroreduction?
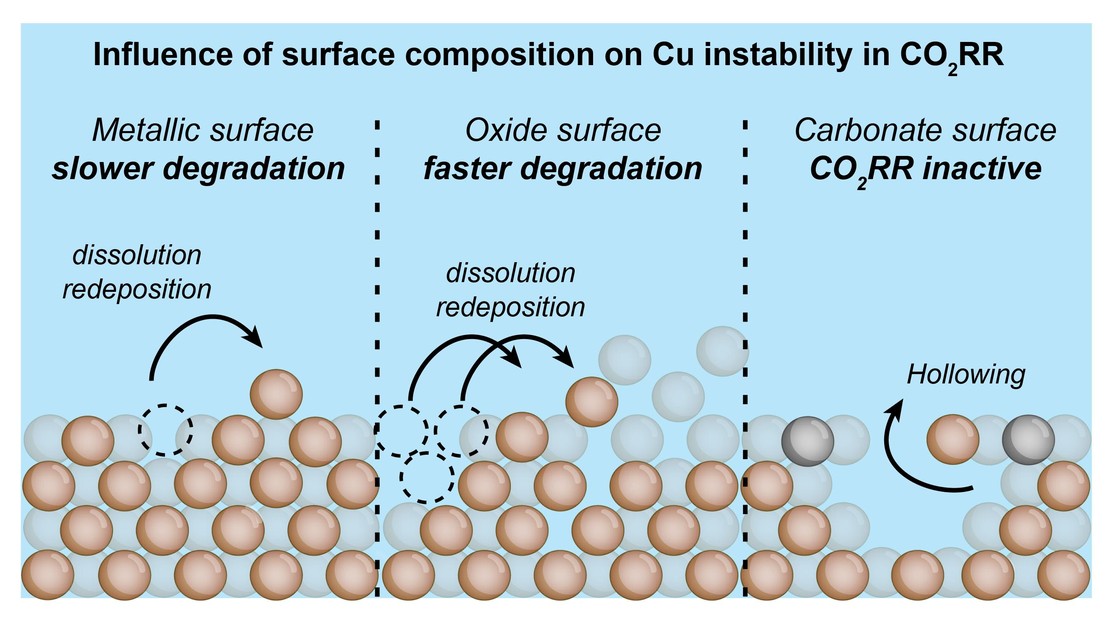
Herein, we investigate the impact of the chemical nature of the initial Cu surface, particularly oxidation state and carbonate formation, on the structural and operational stability of Cu catalysts along with the reconstruction kinetics of the catalyst. We combine state-of-the-art well-defined catalysts with quasi-operando electrochemical liquid-phase transmission electron microscopy (ec-LPTEM) in collaboration with the Tileli group here at EPFL along with electrochemical characterization to learn about underlying differences.
We demonstrate that catalysts with higher initial oxide content undergo faster structural reconstruction and suffer from faster operational deactivation. Interestingly, we find that Cu carbonates further exacerbate structural instability while also suppressing the CO2RR activity.
Our results highlight the critical role of oxides and carbonates in dictating the reconstruction pathways and durability of Cu under CO2RR conditions, offering insights into tuning the Cu-based catalyst design for enhanced CO2RR stability and efficiency.
Read more here!
This publication was created as part of NCCR Catalysis, a National Centre of Competence in Research funded by the Swiss National Science Foundation.
Oxides and Carbonates Accelerate Copper Instability in CO2 Electroreduction. J. Am. Chem. Soc. 2026, 148, 9, 9784–9792