Anti-cancer drug gets a boost when combined with antirheumatic
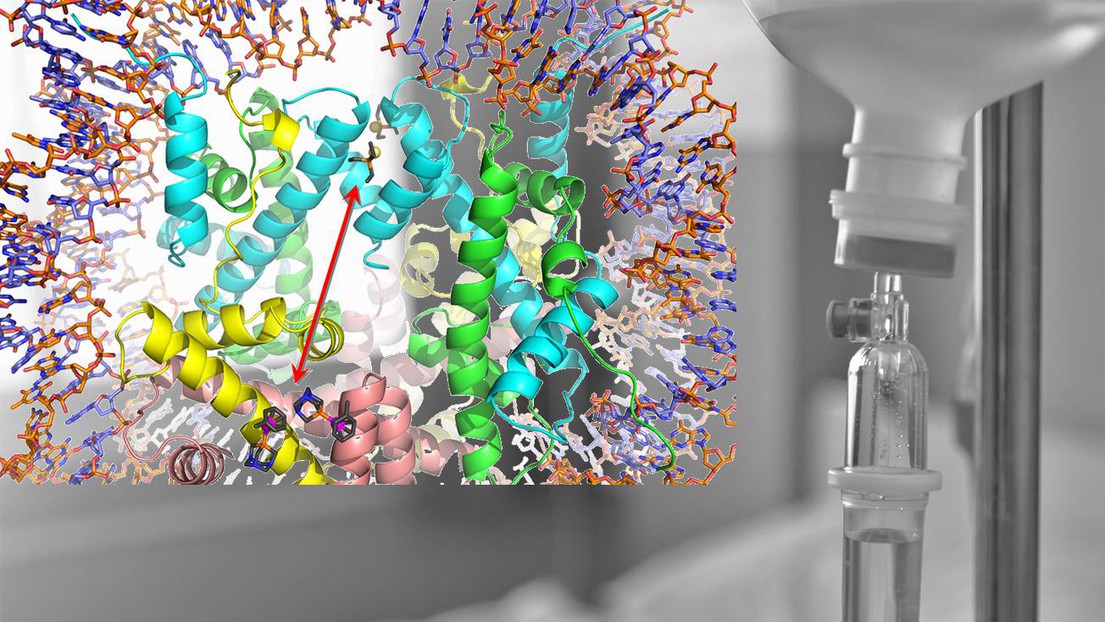
Auranofin (top) and RAPTA-T (bottom) binding different sites of a nucleosome © P. Dyson/EPFL (background: iStock)
Scientists at EPFL and NTU have discovered that combining an anticancer drug with an antirheumatic produces improved effects against tumors. The discovery opens a new path for drug-drug synergy.
One of the goals in pharmacology is to increase the efficiency of drugs by minimizing their side effects. Recently, this effort has led to combining unrelated drugs to exploit their synergistic effects. This “drug-drug synergy” relies on interactions between the individual biological pathways on which each drug acts. Scientists at EPFL and Nanyang Technological University (NTU) have now discovered a synergistic effect between an anticancer and an antirheumatic drug, improving the former’s ability to kill off cancer cells. The work is published in Nature Communications.
The labs of Paul Dyson and Ursula Röthlisberger at EPFL, together with the lab of Curtis Davey at NTU, explored the synergistic effects of two unrelated drugs: auranofin (Ridaura), a gold-containing drug that is used to alleviate the symptoms of rheumatoid arthritis, and RAPTA-T, a ruthenium-containing anticancer drug that disrupts both tumor growth and metastasis, while also reducing the side effects of chemotherapy due to its low toxicity.
Although the two drugs are used for different conditions, auranofin has recently been discovered to also act against cancer. The reason is that, often, drugs do not only bind a single site on a specific molecule, but can also bind and affect other, unrelated sites — either on the same molecule or on a different one. For example, a drug that is meant to bind and activate a receptor could also bind and block an enzyme. This off-site activity frequently gives rise to drug side effects, but separate drug-binding sites can also work together synergistically in a productive fashion.
The researchers looked at the synergistic effects of the two drugs on packaged DNA inside cancer cells. Despite popular depictions, the long strands of DNA in the cell spend most of their time tightly wound around specialized proteins called histones. Whenever a particular sequence, e.g. a gene, is needed, that section of DNA is unwound and read by the appropriate biological machinery.
The study found that combining the two drugs had an increased effect of killing of cancer cells, while individually, the drugs have considerably less impact on cell viability. When RAPTA-T is given, it forms what are known as “adducts” with the histone proteins that package DNA. These adducts disrupt the normal function of DNA and cause the cell to die. In contrast, auranofin is much less prone to form adducts with the histone proteins, unless the two drugs are used together.
The researchers found that the binding of auranofin takes place through an allosteric, “action-over-a-distance” mechanism within the nucleosome, which is the component that contains the cell’s packaged DNA. Here, the researchers discovered that RAPTA-T helps the other drug’s ability to form histone adducts by binding on distant histone sites.
The authors conclude that this newly discovered allosteric mechanism “suggests that allosteric modulation in nucleosomes may have biological relevance and potential for therapeutic interventions.”
This work involved a collaboration of EPFL’s Institute of Chemical Science and Engineering (ISIC) with Nanyang Technological University’s School of Biological Sciences and Institute of Structural Biology. It was funded by the Singapore Ministry of Health National Medical Research Council and Ministry of Education Academic Research Fund Tier 3 Programme, the Swiss National Science Foundation, and NCCR Chemical Biology.
Reference
Zenita Adhireksan, Giulia Palermo, Tina Riedel, Zhujun Ma, Reyhan Muhammad, Ursula Rothlisberger, Paul J. Dyson, Curt A. Davey. Allosteric cross-talk in chromatin can mediate drug-drug synergy.Nature Communications 30 March 2017. DOI: 10.1038/ncomms14860