A high-precision instrument for ophthalmologists
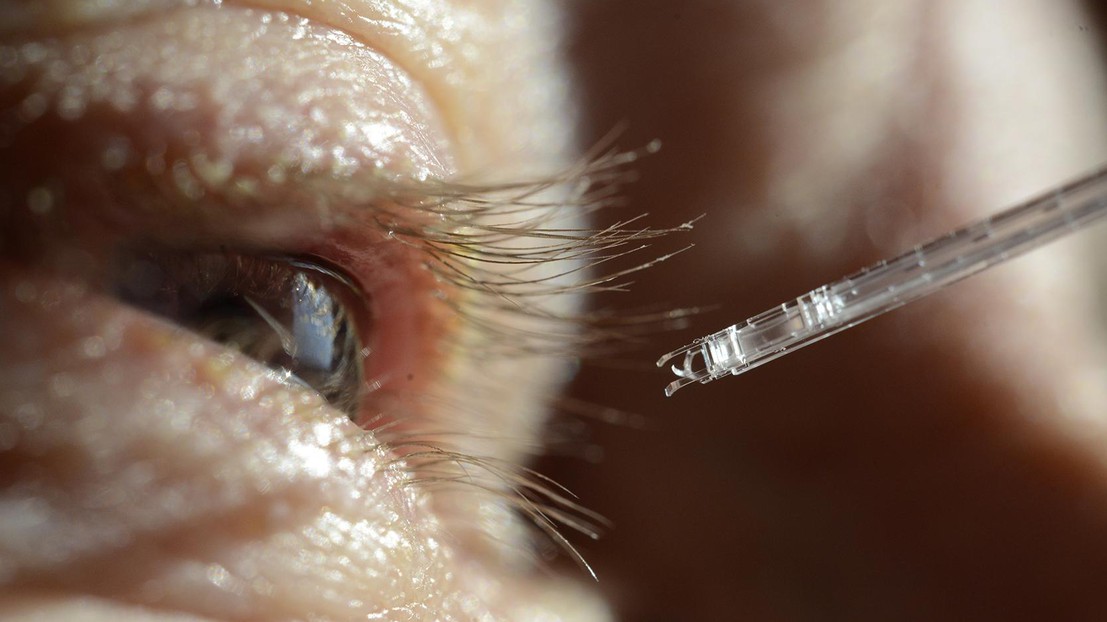
This new microscopic instrument provides the precision and stability needed to treat retinal venous occlusions. © Instant-Lab
EPFL scientists have helped develop a microscopic glass device that doctors could use to inject medicine into retinal veins with unprecedented accuracy. Their instrument meets an important need in eye surgery, delivering exceptional stability and precision.
A team of researchers presented a breakthrough device for eye surgery at EPFL Neuchâtel’s Research Day on 11 September. The device – called SPOT-RVC, which is short for Safe Puncture Optimized Tool for Retinal Vein Cannulation – was developed through a joint R&D project involving two EPFL Neuchâtel labs (Instant-Lab and Galatea) from the School of Engineering, the Jules-Gonin Hospital of Ophthalmology in Lausanne and Ticino-based FEMTOprint. The team’s findings were recently published in the Journal of Medical Devices.
SPOT-RVC is a high-precision, miniaturized medical device made entirely of glass. It’s just 6 cm long and 1 mm thick, and it contains a tiny fluidic channel no wider than a strand of hair as well as a sophisticated mechanism of flexible blades. Doctors can use the device to inject medicine directly into a patient’s retinal veins – something that has never before been possible.

The high-precision miniaturized medical device, SPOT-RVC © Instant-Lab
“We wanted to develop a surgical method for treating retinal vein occlusion, which occurs when the main vein carrying blood away from the eye is blocked. There is currently no way to treat this condition – we can only treat the resulting complications,” says Professor Thomas J. Wolfensberger, the chief physician at Jules-Gonin Hospital. And those complications can be severe. When the retinal vein is blocked by a blood clot, this reduces the amount of oxygen carried to the retina and can trigger sudden vision loss. Over 16 million people around the world suffer from this condition, which mostly afflicts the elderly.
Combining microengineering and microfluid mechanics
Thanks to SPOT-RVC, doctors will be able to inject blood-clot-dissolving compounds directly into patients’ retinal veins safely, without damaging the surrounding tissue. “One of the biggest problems we faced is that because veins are so small and their walls so thin, it’s hard to get the needle into the vein without overpuncturing. It’s like if you want to drill a hole into a plank of wood but don’t want the hole to go all the way through,” says Dr. Charles Baur, a senior scientist at Instant-Lab.
The researchers therefore drew on Instant-Lab’s expertise in flexible microstructures and multistable systems to engineer a microscopic device (< 1 mm in diameter) that can transition from one stable state to another very quickly – in around a millisecond – and in a controlled manner. “With our dynamic perforation mechanism that controls both the penetration force and direction of the needle, retinal veins don’t have time to deform. In addition, the penetration force is independent of the force exerted by the surgeon’s hand, which limits the risk of overpuncturing,” says Dr. Baur.
Another innovative feature of SPOT-RVC is its microscopic, flexible channel that extends all the way down to the needle tip, enabling doctors to inject the medicine. The channel was developed using the advanced microfluids research being carried out by scientists at Galatea. And finally, the device is made of a single piece of fused silica (SiO2). “Since it’s monolithic, there’s no assembly required – a step that would be nearly impossible and would make it very difficult to sterilize the instrument,” says Dr. Baur. The monolithic structure was also developed by Galatea and was fabricated by FEMTOprint, using the company’s high-precision 3D printing method and ultra-rapid laser.
Winner of the Swiss high-precision industry award
FEMTOprint presented SPOT-RVC at the Swiss high-precision industry convention (EPHJ), which was held this past June in Geneva. The device won the 2019 Exhibitors’ Grand Prix – an encouraging start.
For now the device is still in the prototyping stage. “We got good results from our in vitro and in vivo tests,” says Dr. Baur. “Now we need to conduct preclinical trials and obtain the necessary certifications. Then we’ll move on to the production stage, which will require a fairly large investment. I genuinely hope that one day the device will become a useful tool for eye surgeons.”
Discover the mechanism in video on https://www.youtube.com/watch?v=1ZNGuvkzNsE&feature=youtu.be.
Zanaty, M. et al. “Programmable Multistable Mechanisms for Safe Surgical Puncturing,” ASME. J. Med. Devices. 2019;13(2):021002-021002-10. doi:10.1115/1.4043016.



