A breakthrough in our understanding of ionic liquids

Electrolytes are usually conducting media, in which charged ions move freely in a solvent. Now an experiment led by Professor Jacques-E. Moser has revealed a new mechanism for electrical conduction in ionic liquid electrolytes.
In a recent paper of ChemPhysChem, Professor Moser’s team has demonstrated the existence of a new conduction mechanism that does not involve the transport of ions. This discovery was made while looking for ways to improve the conductivity of ionic liquids, which are used as electrolytes in dye-sensitized solar cells. The peculiarity of these liquids is that they contain only ions and no neutral solvent molecule. Evaporation of the liquid and its degradation are, thus, prevented.
In the belief that viscosity was limiting the capacity of the medium to let current flow through it, the scientists studied its conduction properties between its solid state, cooled to –93°C, and its liquid state, at room temperature. They were very surprised to discover that more than twenty degrees below the melting point, the conduction was almost identical to that of the liquid. This observation, contrasting with the existing model of mobile electrical charges, demanded further research.
The phenomenon was therefore studied in more details by applying terahertz time-domain spectroscopy. The resulting measurements enabled the detection of the vibrations of the ions, as well as their way to associate with each other. This achievement was made possible thanks to ultra-short laser pulses that generate THz radiation, which lies between the infrared and the microwave frequency range. This radiation has the property of interacting with mobile electrical charges and low-frequency vibrations of the matter.
It was thus possible to observe in an ionic liquid, containing iodide and triiodide ions, a bond-exchange phenomenon. In this scenario, an iodide ion (I-) binds to a neighboring triiodide (I3-) ion, while at the other end of the chain a bond is broken and a new I- ion is liberated. This mechanism governing the displacement of electrical charges only implies back-and-forth movements over very short distances, of the order of 3 angstroms, and does not require the transport of ions over longer ranges. Thanks to the bond-exchange between ions, electrical conduction remains efficient, even in viscous liquids and at very low temperature.
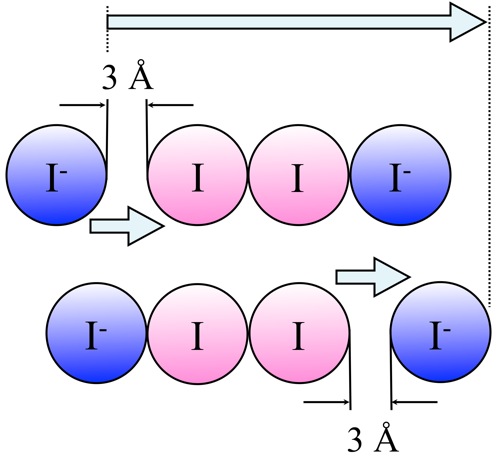
A similar mechanism was suggested by Theodor Grotthuss at the beginning of the 19th century to explain the conductivity of water, but had never been demonstrated for an ionic liquid.
This breakthrough opens up a new field of research. It paves the way towards interesting and innovative applications, for instance in the design of more stable and efficient third-generation solar cells and of new high-capacity batteries.
Links:
http://photochemistry.epfl.ch/reprints/CPC121.pdf
http://gdp.epfl.ch