A 3D printable scaffold to support fast bone growth
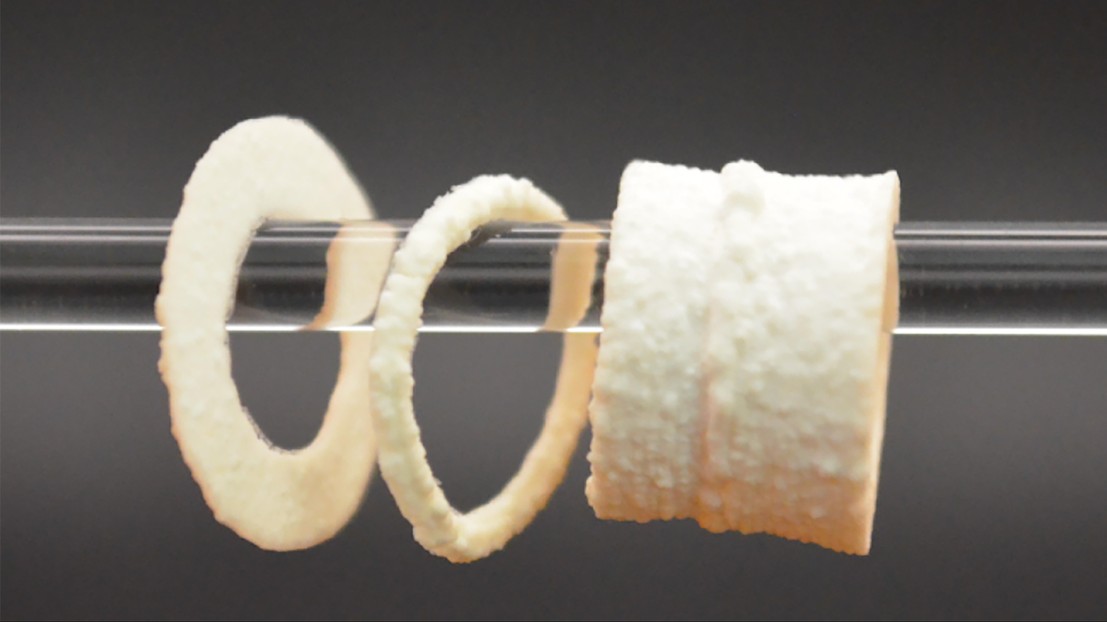
Cylindrical demonstrations of the 3D printed scaffolds. 2026 EPFL SMaL CC BY SA
A bone-like composite developed at EPFL, in collaboration with researchers from ETH Zurich, Empa and the University of Fribourg, uses naturally occurring enzymes to accelerate mineralization through an energy-efficient, room-temperature process. The strong, lightweight material shows promise for bone repair applications.
Inspired by the resilient and self-repairing mechanical properties of bone, scientists have been developing synthetic materials using one of bone’s main components: a mineral called hydroxyapatite (HA). However, the high-temperature processes typically required to produce HA-based materials use significant energy and restrict the use of biologically active components, like enzymes, to promote bone growth.
Researchers in the Soft Materials Laboratory (SMaL) in EPFL’s School of Engineering, in collaboration with ETH Zurich, Empa and the University of Fribourg, have now developed a way to 3D-print HA-based scaffolds using a room-temperature process that harnesses enzymes for fast mineralization. The resulting bone-like porous scaffolds can become load bearing within just 7 days. The research has been published in Advanced Functional Materials.

“Our idea was to generate a 3D printable and injectable ‘ink’ that can be mineralized into scaffolds with mechanical properties similar to those of highly porous trabecular bone, which is found in human vertebrae and the ends of long bones like the femur,” says laboratory head Esther Amstad. “We hope that our technology’s combination of mechanical performance, bioactivity, and energy-efficient processing will open new avenues for bone tissue engineering.”
Engineering space for bone remodeling
The EPFL team’s ‘ink’ is produced by embedding the enzyme alkaline phosphatase into gelatin microparticles and incubating them in a solution with calcium and phosphate ions. The enzyme triggers the formation of HA crystals that stiffen and strengthen the printed scaffolds. After only four days of mineralization, the composite can bear the average weight of an adult human on an area as small as 1.5 cm x 1.5 cm.
The scientists also add enzyme-free gelatin microfragments, which melt when the scaffold is incubated, leaving pores behind. After implantation at the site of a bone fracture, for example, these pores can be replaced by healthy cells to promote the growth of new bone. Importantly, tuning the density of these microfragments allows the team to control the scaffold’s porosity. By introducing pores that make up around 50% of the scaffold volume, the researchers create plenty of room for cells to infiltrate and remodel the scaffolds, opening up new possibilities for natural bone regeneration.
Our work might lay the foundation for injectable scaffolds that aid bone regeneration.
In one experiment, 14 days after seeding scaffolds with human stem cells and placing them in a bone growth-supporting medium, the researchers detected the presence of collagen and the bone matrix protein osteocalcin – both indicators of cell growth. These results support the approach’s potential for tissue engineering.
Amstad adds that the SMaL team’s enzyme-aided approach yields HA scaffolds that are stronger than those produced via high-temperature methods, demonstrating compressive strength comparable to that of human trabecular bone. Their technique can be used to print highly complex scaffolds and can also be used with commercially available bioprinters.
“Looking ahead, our work might lay the foundation for injectable scaffolds that aid bone regeneration and potentially enable patients to load their broken bones much earlier than can be achieved with currently available technologies,” she says.
F. Bono, A. Puiggalí-Jou, G. Cocchi, et al. “ 3D-Printed Porous Hydroxyapatite Formed via Enzymatic Mineralization.” Advanced Functional Materials (2026): e26568. https://doi.org/10.1002/adfm.202526568

